A new study could lead to new ways to treat movement disorders like those associated with Parkinson’s disease. Credit: Angela Cenci Nilsson
New research has shed light on the interaction between the start and stop functions of the brain that may lead to treatments for disorders like Parkinson’s disease.
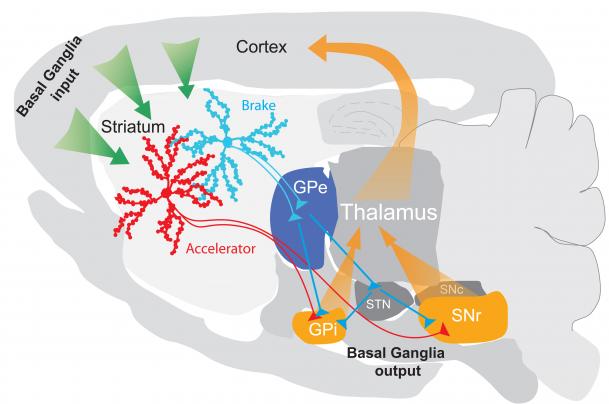
Researchers at the Lund University in Sweden have presented new research on the interaction between the “accelerator” and the “brake” in the striatum, the part of the brain that controls movements.
Parkinson’s disease begins to slow and stiffen patients’ movements in the early stages and patients are often given L-dopa as treatment, but after a few years of treatment patients often exhibit uncontrolled jerking motions called dyskinesias.
Other diseases, including Huntington’s disease and several hereditary conditions, are also associated with similar movement disorders.
Parkinson’s researcher Angela Cenci Nilsson, from Lund University said the role of the striatum in movement disorders has been contested in the science community.
“We know that the striatum plays an important role in movement control,” Nilsson said in a statement. “But which neural pathways are most important has been hotly debated.”
The striatum has two principal types of cells that form distinct neural pathways called “direct pathway” and “indirect pathway.”
Scientists have often debated whether both pathways are equally important in all situations and whether they need to cooperate or can work independently.
The Lund researchers were able to apply a method called chemogenetics— using a harmless virus to introduce a new gene into striatal cells in laboratory mice. All the experimented animals had a “Parkinson’s-like injury,” both with and without dosages of L-dopa.
The gene was coded for the production of a receptor protein activating the relevant neural pathway, but the receptor became stimulated only when the mouse was administered a particular substance that lasted a couple of hours.
The scientists were able to use this method to control the activity of cells forming the direct or indirect pathway while studying the animals’ behavior.
The results showed that all types of movements were controlled by both pathways.
In the mice treated with L-dopa, activation of the direct pathway produced faster movements but also more severe dyskinesias.
“We interpret these results to mean that the pathways need to interact in all situations, even in Parkinson’s-like conditions and upon L-dopa treatment,” Nilsson said. “You can’t have only acceleration and no braking, but must instead balance both functions in a precise manner.”
Nilsson said that L-dopa therapy gives complications when it “inactivates the brake” by strongly inhibiting the indirect pathway, while “pressing too hard on the accelerator” by overstimulating the direct pathway where dyskinesias can occur.
The new findings could help explain why it has been challenging to develop new drugs for Parkinson’s disease. Drug development has often focused on one or the other neural pathway, while Nilsson’s study suggests a need to intervene on both pathways using either a drug that regulates both of them or two complementary drugs.
“Our results could be of great significance both for basic research and for therapeutic research,” Nilsson said.
The study was published in The Journal of Clinical Investigation.
Filed Under: Drug Discovery




